Happy Valley Permanent Vegetation Plots
Walker, D.A., Auerbach, N.A., Nettleton, T.K., Gallant, A., Murphy, S.M. 1997. Happy Valley Permanent Vegetation Plots: Site factors, physical and chemical soil properties, plant species cover, photographs, soil descriptions, and ordination. Arctic System Science Flux Study, Institute of Arctic and Alpine Research, University of Colorado, Boulder, CO.
- R4D Program Data Report (Data Report: PDF)
- Introduction
- Methods
- Ordination
- Acknowledgements
- References
- Appendices
- Appendix A. Field descriptions of soils - PDF - MS Word
- Appendix B. Plot and soil photos - PDF
- Figures
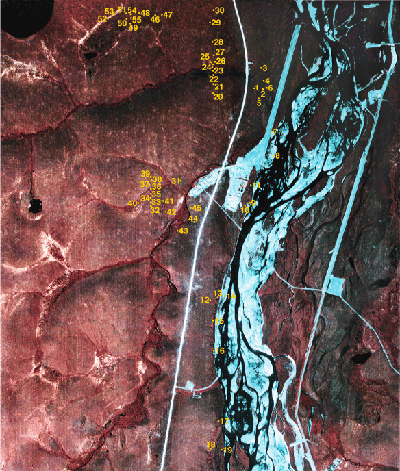
- Figure 1. Relevé locations
- Figure 2. Detrended Correspondence Analysis
- Tables
- Table 1. Preliminary vegetation classification based on habitat and dominant species and sample numbers for each type - PDF - MS Excel
- Table 2. List of vegetation communities and sites sampled in 1994 - PDF - MS Excel
- Table 3. Sample site description data sheet including legend for environmental variables - PDF - MS Excel
- Table 4. Sample relevé data sheet including the Braun-Blanquet cover scale - PDF - MS Excel
- Table 5. Environmental data collected from Happy Valley - PDF - MS Excel
- Table 6. Relevé size, percent live cover and non-living cover and height of vegetation - PDF - MS Excel
- Table 7. Soils data from Happy Valley relevés - PDF - MS Excel
- Table 8. Happy Valley relevé species data - PDF - MS Excel
- (see Tables 1, 2, 4, 5, 6, 7, and 8 for links to individual plots; plant species name links to a descriptive webpage with photos)
Introduction
This data report is a summary of environmental, soil and vegetation information collected from 55 relevé plots at the Happy Valley research site along the Sagavanirktok River, located at 69˚14’N, 148˚50’W in northern Alaska. Figure 1 shows the location of the relevés at Happy Valley. The report brings together all the available information collected from the plots, as well as offering a basic analysis of the communities. This information can be used to further investigate plant community classification, and environmental relationships.
Methods
Reconnaissance survey, sampling dates, and plot locations
A reconnaissance survey was conducted prior to sampling to define the primary vegetation types of Happy Valley, and a total of 18 vegetation types were defined (Table 1). Formal relevé sampling for the plots in this data report was done during the period July 18-27, 1994. An attempt was made to sample at least three relevés for each of the preliminary vegetation types defined, but this was not always possible. Each relevé was described based on the dominant plant species identified (Table 1 and Table 2).
The relevés were located in homogeneous areas of vegetation using the centralized replicate method of the Braun-Blanquet approach to vegetation classification (Mueller-Dombois and Ellenberg, 1974; Westhoff and van der Maarel, 1978).
All the relevés are permanently marked with a 4-foot black and white-striped 1-inch PVC pipe. The relevé number is stamped into an aluminum tag at the top of the post.
Sampling
Relevé size, species cover estimation, and photographs
The relevés have no fixed size because our main objective was to obtain a complete species list for each relevé.
Photographs were taken of each relevé (see Appendix B). Usually photos were taken of 1) landscape view of the site, 2) close-up of the vegetation, and 3) close-up of the soil profile.
Site factors
The site of each relevé was described according to the variables listed in sample site description sheet (Table 3) plus thaw depth measurements, estimates of cover of bare soil, rocks, the height of vegetation, and the major plant growth forms shown in the sample relevé sheet (Table 4). These data are summarized in Table 5 and Table 6.
Soils
Field sampling
Soil pits were dug adjacent to the relevés and described and classified according to the U.S. soil taxonomy [Soil Survey Staff, 1975]. Soil samples were collected from each horizon and air dried in the laboratory. Bulk density and soil moisture samples were taken from the sides of the soil pits or from large solid plugs for the wet soils using a 240-ml soil can.
Laboratory analysis
Laboratory analyses were conducted at the Colorado State University Soil Testing Laboratory, Fort Collins. The laboratory’s routine analysis was run on all samples [pH (saturated paste); electric current; NO3-N, (KCL extract); P, K, Zn, Fe, Cu, Mn, (NH4HCO3-DPTA extract); lime estimate, texture estimate (by hand); organic matter (Walkley-Black or ash method); and Sodium Adsorption Ratio]. Selected samples were analyzed for percent sand, percent silt and percent clay; and Ca, Mg, Na, K (NH4OAc extract). These methods are described in Page et al. (1982) and Klute (1986). The soils data are in Table 7.
Vegetation
Plant communities were determined and sample plots were marked with stakes. The area surrounding each plot marker was searched until no new species in the plant community being sampled were encountered . Estimates of vegetation cover used the Braun-Blanquet cover-abundance scale (r = rare; + = common but less than 1%; 1 = 1-5%; 2 = 6-25%; 3 = 25-50%; 4 = 51-75%; 5 = 76-100%). Cover abundance values are relatively broad subjective classes, and were determined by estimating cover within the general area of the stake. Voucher collections were made for all vascular plants, bryophytes, and lichens occurring in the relevé. Unknown collections were identified and known collections were verified as follows: Bryophytes by Dr. Olga Afonina; liverworts by Dr. Alexey Potemkin; and lichens by Dr. Mikhail Zhurbenko all at the Komarov Botanical Institute, St. Petersburg, Russia. Table 8 contains the species cover raw data.
Ordination
We used Detrended Correspondence Analysis (DCA) ordination (Hill, M.O. and H.G. Gauch, 1980). Species were weighted equally and detrended by segments. Three aquatic samples (HV-3, HV-46, and HV-48), one wet sedge tundra (HV-9), and one frost scar plot (HV-42b) were excluded from the ordination. These relevés are so dissimilar in species composition that when included, slight differences between other community types were difficult to distinguish in the ordination space. The ordination displays relevés in a two dimensional space according to their species similarity. Environmental variables were related to the ordination axes with a biplot diagram. The biplot diagram indicates the strength of correlations for significantly correlated environmental variables (Dargie, 1984; Jongman et al., 1987).
Results from the ordination capture three significant environmental variables that affect variation in the vegetation composition, stability, exposure and soil moisture (Figure 2). Relevés that occur in preliminary classifications also occur in recognizable clusters in the ordination space.
Acknowledgements
This work was funded by the Arctic System Science Flux Study, National Science Foundation grant OPP-9318530. The Colorado State University Soil, Water and Plant Testing Laboratory provided soil analysis. William Gould, Mike Mantassa, and Andrew Lillie assisted with the preparation of this report.
References
- Dargie, T.C.D. 1984. On the integrated interpretation of indirect site ordinations: a case study using semi-arid vegetation in southeastern Spain. Vegetatio. 30:15-32. . doi:10.1007/BF00039980.
- Hill, M.O., Gauch, H.G. 1980. Detrended correspondence analysis, an improved ordination technique. Vegetatio. 42:47-58.
- Jongman, R.H.G., ter Braak, C.J.F., van Tongeren, O.F.R. 1987. Data Analysis in Community and Landscape Ecology. Pudoc. Wageningen. 299 .
- Klute, A., Page, A.L. 1982. Methods of soil analysis. American Society of Agronomy: Soil Science Society of America. Madison, WI. 2nd ed. . - WorldCat.org listing
- Mueller-Dombois, L.D., Ellenberg, H. 1974. Aims and Methods of Vegetation Ecology. John Wiley and Sons. New York. 547 .
- Page, A.L., Miller, R.H., Keeney, D.R. 1982. Methods of soil analysis, Part 2. Chemical and microbiological properties. American Society of Agronomy, Inc. and Soil Science Society of America, Inc. Madison, WI. 1159 .
- Staff, U.S.T.S.S. 1974. Soil taxonomy of the National Cooperative Soil Survey. Soil Conservation Service. Washington, D.C. 754 .
- Walker, D.A., Lederer, N.D., Walker, M.D. 1987. Permanent vegetation plots (Imnavait Creek): site factors, soil physical and chemical properties, and plant species cover. R4D Program Data Report. 43 pp. PDF
- Westhoff, V., van der Maarel, E. 1978. The Braun-Blanquet approach. Classification of Plant Communities. pp. 287-399. PDF



